—
#DIYPS=on path to artificial pancreas but not limited to those using newest diabetes tech. http://bit.ly/1mMS7LA @danamlewis @scottleibrand
—
Like many others, we’ve been reading the latest in the New York Times about the impact of diabetes technology innovation on the cost of managing the disease – not to mention the reactions to the piece, the response to the reactions, the reactions to that, etc.
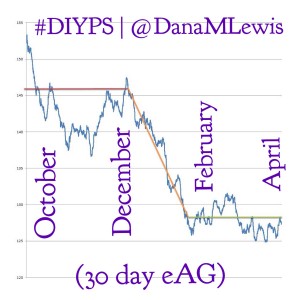
We believe there is a better way forward, and #WeAreNotWaiting to make it happen. Innovation can happen in a low-cost way, and can be scaled to support a broad patient audience, without contributing to or requiring significantly increased healthcare costs. #DIYPS for example (check out these results) was developed by two individuals, not an organization, with the goal of solving a well-known problem with an existing FDA-approved medical device. As recounted here (from Scott) and here (from Dana), we set out to figure out a way to augment continuous glucose monitor (CGM) alerts, which aren’t loud enough to wake heavy sleepers, and to alert a loved one if the patient is not responding.
We were able to solve those problems, and include additional features such as:
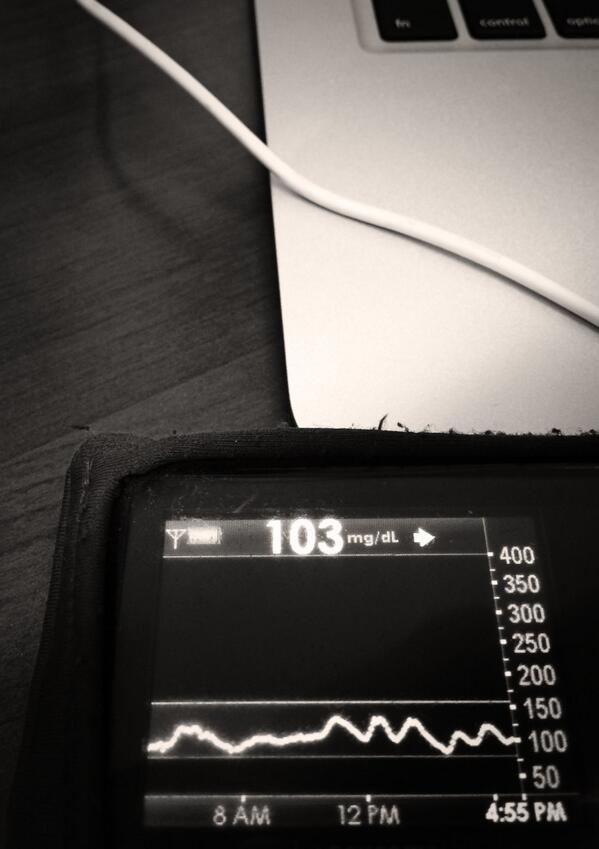
- Real-time processing of BG, insulin on board, and carbohydrate decay
- Customizable alerts based on CGM data and trends
- Real-time predictive alerts for future high or low BG states (hours in advance)
- Continually updated recommendations for required insulin or carbs
While #DIYPS was invented for purposes of better using a continuous glucose monitor (CGM) and initially tailored for use with an insulin pump, what we discovered is that #DIYPS can actually be used with many types of diabetes technology. It can be utilized by those with:
- CGM and insulin pump
- CGM and multiple daily injections (MDI) of insulin
- no CGM (fingerstick testing with BG meter) and insulin pump
- no CGM (fingerstick testing with BG meter) and multiple daily injections (MDI) of insulin
We think this type of device-agnostic software/technology is critical as we work on pathways to the artificial pancreas systems (APS). While we hope APS is out on the market soon (10 years ago would’ve been nice :)), we know it will take several years to a decade. And, even when it comes out, APS will be expensive. It may not be covered by insurance. Even with insurance, people may not be able to afford it. And even if everyone could afford it, some people may prefer not to use it.
—
We believe technology like #DIYPS can, and must, scale to take advantage of real-time data from CGMs, insulin pumps, and any other new diabetes technology, and help patients achieve the best possible health and quality-of-life outcomes while waiting for APS systems to become available. But at the same time, we want to build these types of tools so that anyone with any combination of diabetes tools can use them to better self-manage their own particular condition. For example, availability of bolus calculator tools is often limited to those with pumps. #DIYPS can be used as a simple bolus calculator, with the added benefit that it can keep track of carb absorption and allow the user to calculate correction accurate boluses while still digesting a meal. Packaged into a simple web or app interface, this would allow people to do the same type of quick data input and calculations to be able to verify/support their mental math.
While #DIYPS is a very effective prototype, we don’t expect it to be the only interface that everyone with Type 1 diabetes uses. Rather, we hope to integrate it with projects like Tidepool that will allow anyone, with any kind of meter, pump, or CGM, to easily upload their data, and then use any number of tools like #DIYPS to interact with their own data and get both real-time and historical insights from it that will let them improve their own diabetes self-care. However, to make this possible, we need all medical device makers to open up their devices to allow patients real-time programmatic access to their own data. (A good example – there’s no access to temp basal history on Medtronic pumps!)
We need people and companies with innovative ideas to focus on making those ideas available as device-agnostic software, not solely as a proprietary feature on a single non-interoperable medical device. And most of all, we need everyone to focus on making the fruits of innovation available as widely as possible, even to patients without the financial resources to buy cutting-edge hardware.
After all, #DIYPS is proof that low-cost innovation can have big results.
Recent Comments